|
The current versions of Hidrex Iontophoresis machines do not contain colour coded cables, ports or electrodes. However, recent actual studies have shown that the red positive anode side will respond better than the negative cathode on modern machines. There wasn’t too much emphasis placed on this switching action at that time, as this effect was not clinically proven – the evidence was purely anecdotal. In these instances, and for these machines, it was recommended to swap sides so that you can even up the responses and the general advice was to alternate which side was treated by the positive red or negative black side. This is because there is slightly more ionic activity on the positive anode so theoretically, if you only ever treated one side of the body with the positive side, you may find that side gains better results sooner. Knowing this information was important at that time as there was thought to be a slightly (in most cases unnoticeable) better response rate at the positive (Red +) side. This colour coding allowed you to plug in the corresponding cables and know visually, which side of the body was being treated with the positive and negative sides. In addition to this, the electrode plates supplied on these models were also colour coded so it is little wonder people felt it was an important part of their iontophoresis treatment. On the back of the control panels on these iontophoresis machine, the cable ports were also colour coded. It also doesn’t help that previous Hidrex models, such as the Hidrex PSP1000 and the Hidrex GS400 were supplied with colour coded cables. To help clear this up, the following explains which is which:Ĭathode or Black = Negative (-) Previous Hidrex Iontophoresis Models (PSP100 and GS400) 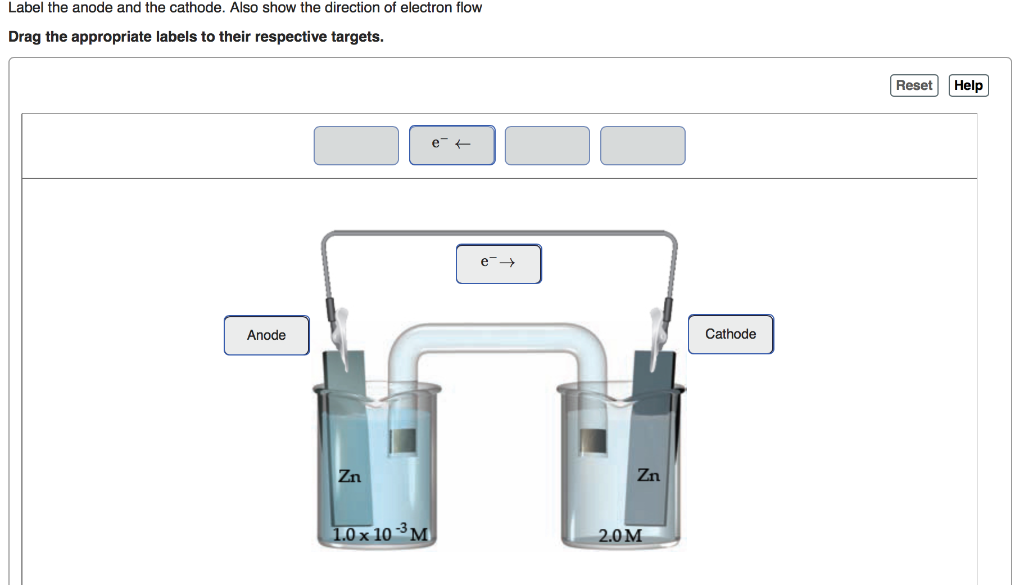
It doesn’t help that each side can be referred to as either red or black, positive or negative or anode and cathode! In fact, zinc would oxidize completely before iron would begin to react.Iontophoresis cables and polarity changes explained Like the title of this post indicates, it is fair to say that there is a fair bit of confusion surrounding the positive and negative cables and where they need to be in relation to your iontophoresis machine, and how this affects your treatment of Hyperhidrosis. This difference in reduction potential means that Zinc would oxidize much faster than iron would. The standard reduction potential of iron is about -0.44 volts. According to the table of Standard Reduction Potentials, the standard reduction potential of zinc is about -0.76 volts. The addition of zinc, a sacrificial anode, would prevent the iron metal from "corroding". Under normal circumstances, the iron metal would react with the electrolytes and begin to corrode, growing weaker in structure and disintegrating. Metal in seawater is one such example with the iron metal coming into contact with electrolytes. Sacrificial anodes are among several forms of cathode protection. It is important to ensure that these metals last as long as they can and thus necessitates what is known as cathode protection. These metal surfaces are used all around us - from pipelines to buildings to ships. Corrosion is the process of returning a metal to its natural state as an ore and in this process, causing the metal to disintegrate and its structure to grow weak. When metal surfaces come into contact with electrolytes, they undergo an electrochemical reaction known as corrosion. The sacrificial anode will be consumed in place of the metal it is protecting, which is why it is referred to as a "sacrificial" anode. Sacrificial Anodes are created from a metal alloy with a more negative electrochemical potential than the other metal it will be used to protect.  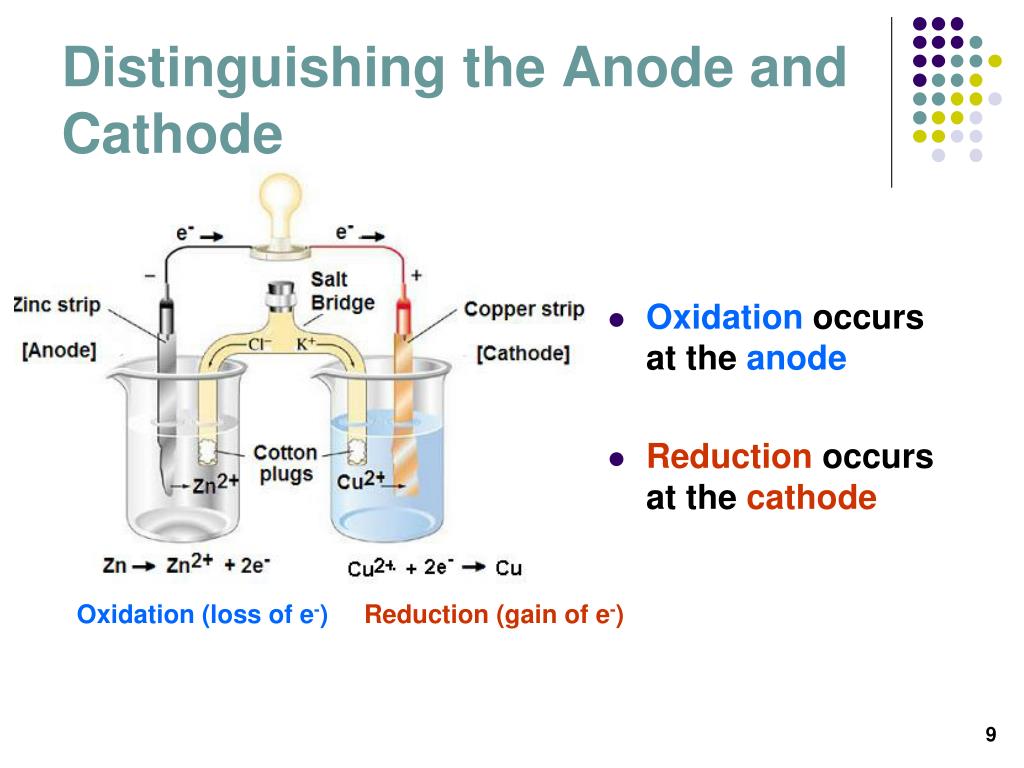
Sacrificial Anodes are highly active metals that are used to prevent a less active material surface from corroding. What materials are used for sacrificial anodes?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
